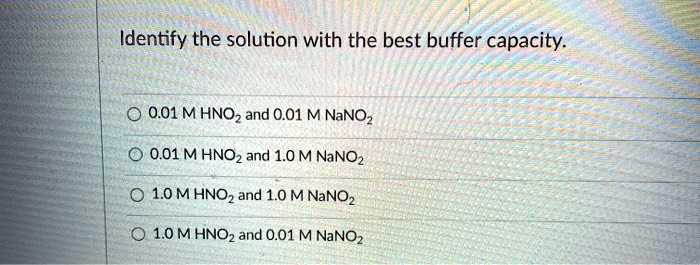
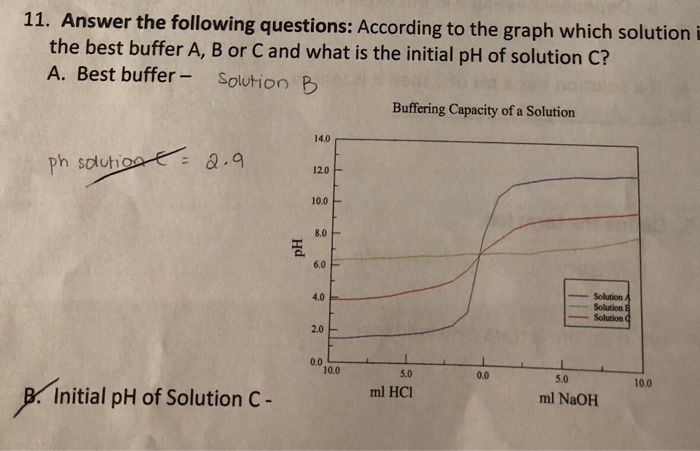
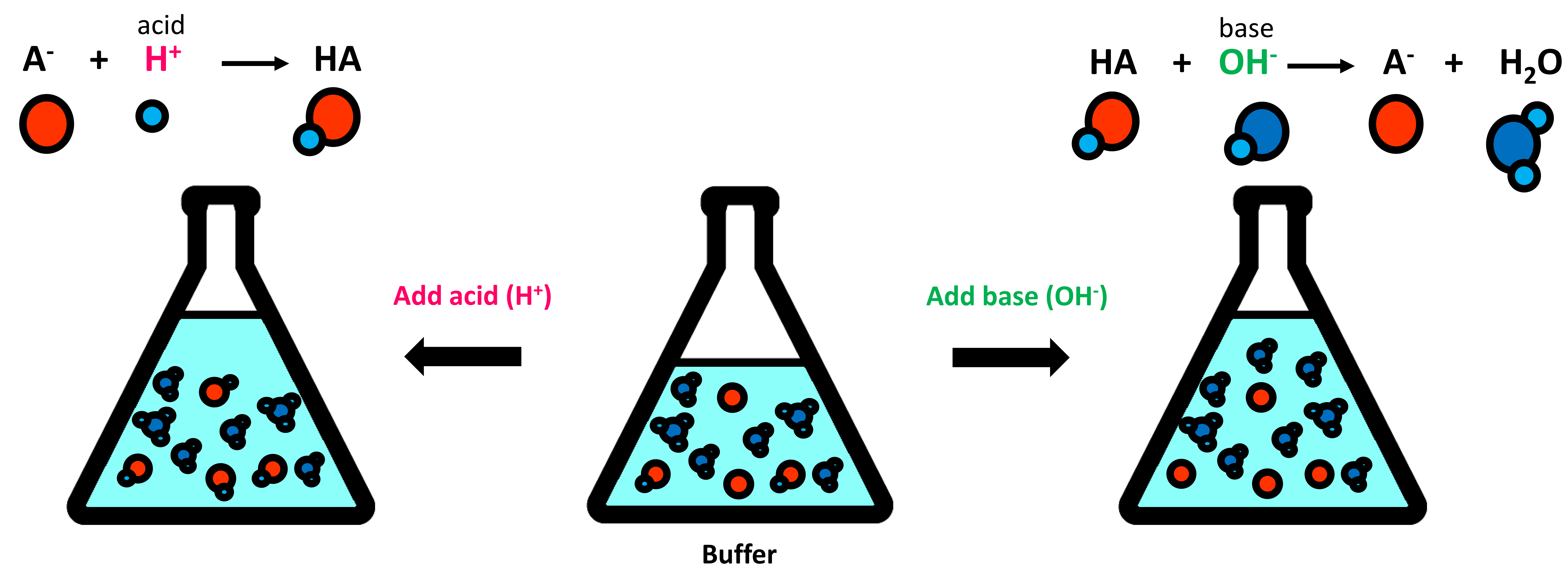
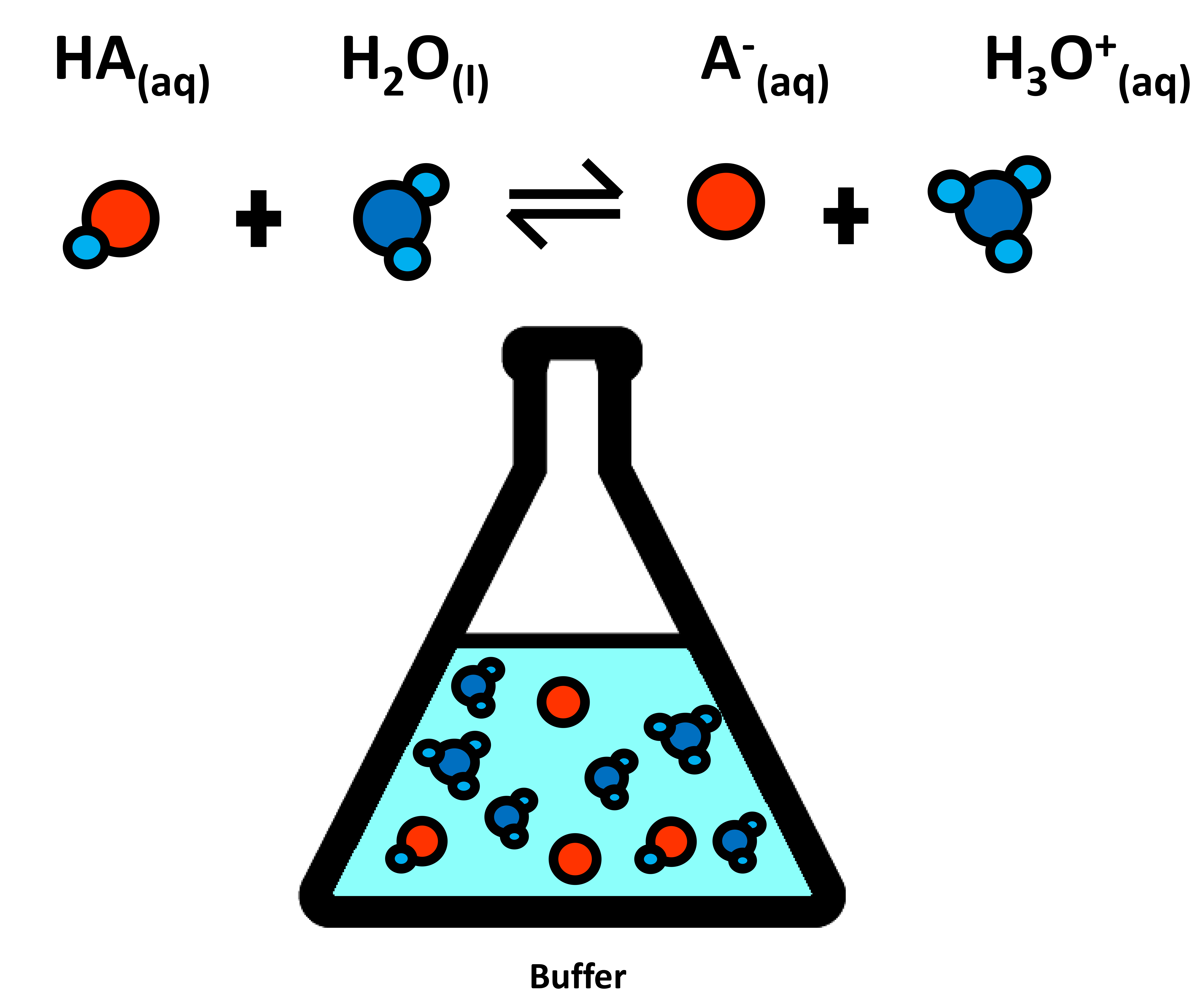
OneClass: Correct The best buffer will be the one whose weak acid component has a pKa closest to the ...

SOLVED: Question 12 Which pair would make the best buffer system? A) weak acid weak conjugate base B) strong acid weak conjugate base C) strong base weak conjugate acid D) strong acid

The Best Car Buffers (Including for Beginners and Professionals, with Dual Action and Variable Speed Polisher Options)